Effectiveness of mRNA vaccine boosters against infection with the SARS-CoV-2 omicron (B.1.1.529) variant in Spain: a nationwide cohort study - The Lancet Infectious Diseases

Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults — Increasing Community Access to Testing ...
Data suggests mRNA booster dose generates stronger antibody response after J&J shot - Axios | Reuters

Boosting with variant-matched vaccines: an opportunity to win the race against Omicron | Signal Transduction and Targeted Therapy

Effectiveness of a bivalent mRNA vaccine booster dose to prevent severe COVID-19 outcomes: a retrospective cohort study - The Lancet Infectious Diseases

Safety Monitoring of COVID-19 mRNA Vaccine Second Booster Doses Among Adults Aged ≥50 Years — United States, March 29, 2022–July 10, 2022 | MMWR

Real-World Association Between mRNA Vaccination and Infection From the Omicron Strain of SARS-CoV-2: A Population-Level Analysis - AJPM Focus

Effectiveness of Bivalent mRNA Vaccines in Preventing Symptomatic SARS-CoV-2 Infection — Increasing Community Access to Testing Program, United States, September–November 2022 | MMWR

Effectiveness of mRNA vaccine boosters against infection with the SARS-CoV-2 omicron (B.1.1.529) variant in Spain: a nationwide cohort study - The Lancet Infectious Diseases

Safety, immunogenicity, and efficacy of the mRNA vaccine CS-2034 as a heterologous booster versus homologous booster with BBIBP-CorV in adults aged ≥18 years: a randomised, double-blind, phase 2b trial - The Lancet

CDC - COVID-19 BOOSTER UPDATE: Everyone ages 18+ is eligible for a COVID-19 booster. If you got an mRNA vaccine, either Pfizer-BioNTech or Moderna, you may get a booster dose 6 months

Immune Memory Response After a Booster Injection of mRNA-1273 for Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) | medRxiv
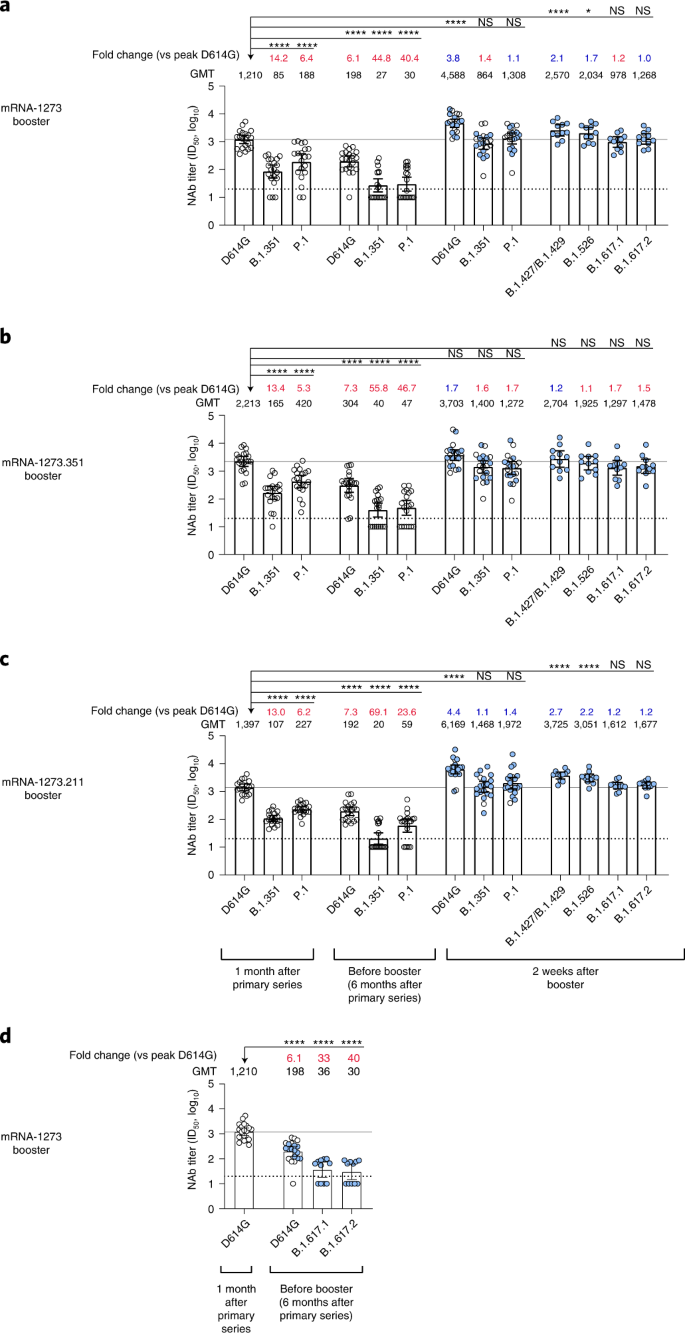
Safety and immunogenicity of SARS-CoV-2 variant mRNA vaccine boosters in healthy adults: an interim analysis | Nature Medicine

Safety and immunogenicity of SARS-CoV-2 variant mRNA vaccine boosters in healthy adults: an interim analysis | Nature Medicine
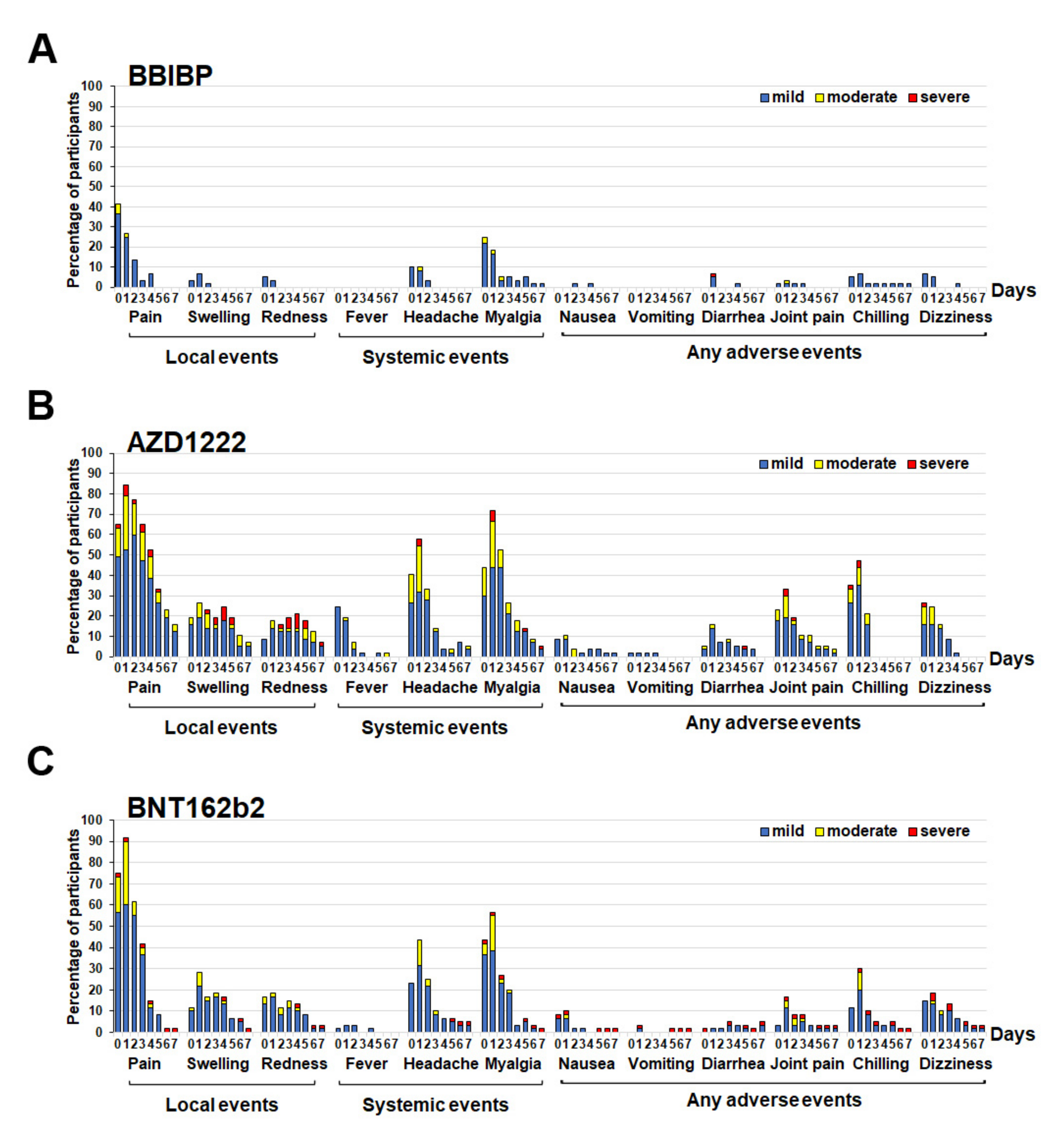
Vaccines | Free Full-Text | Safety and Immunogenicity of the Third Booster Dose with Inactivated, Viral Vector, and mRNA COVID-19 Vaccines in Fully Immunized Healthy Adults with Inactivated Vaccine