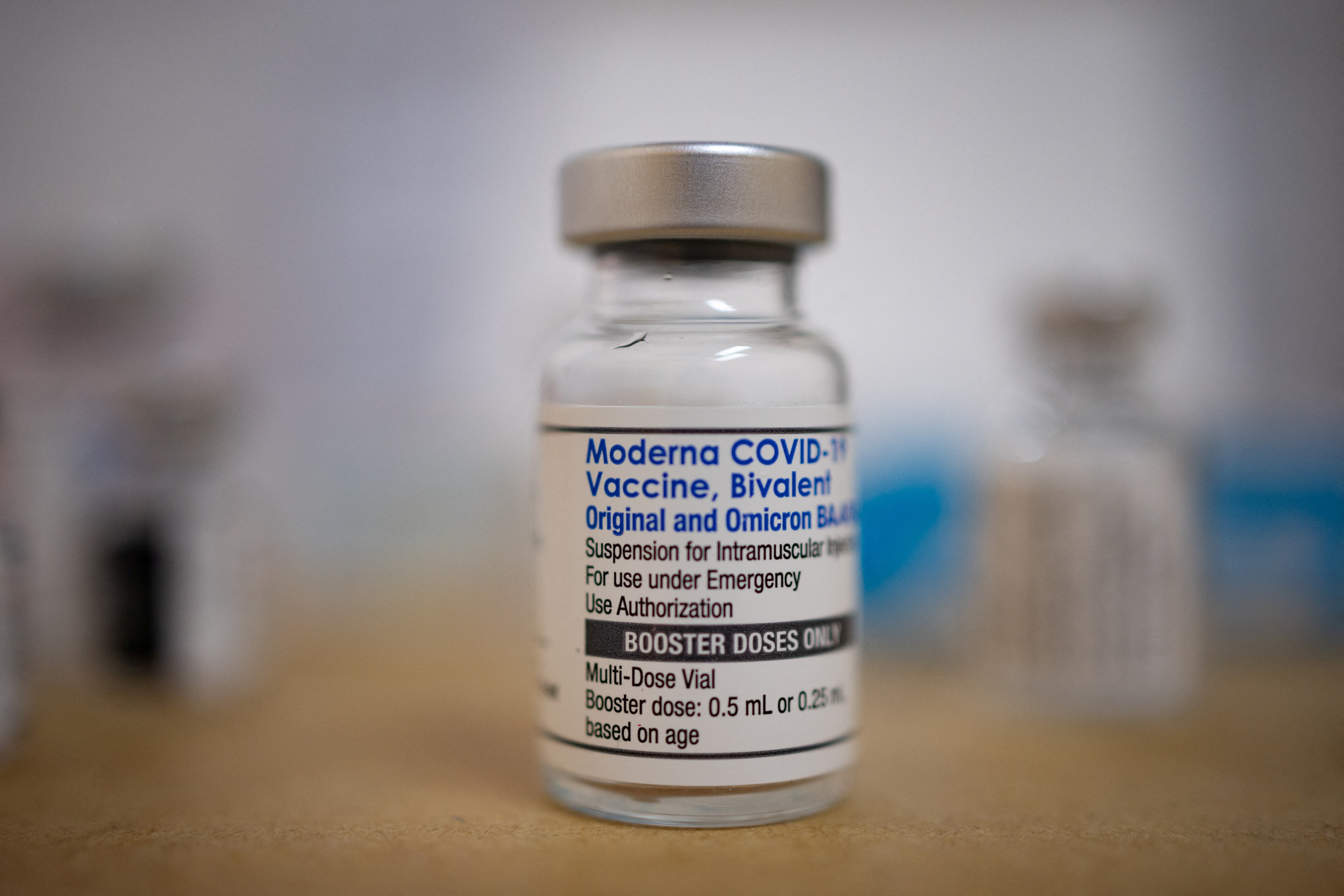
Moderna on X: "@GovCanHealth has authorized the use of our Omicron-targeting #bivalent COVID-19 booster #vaccine, mRNA-1273.214, as a booster dose for active immunization against #COVID19 in individuals 18 years of age and
Bivalent COVID-19 booster vaccine receives MHRA approval - Hospital Pharmacy EuropeHospital Pharmacy Europe
Common Questions About Bivalent COVID-19 Boosters | Johns Hopkins | Bloomberg School of Public Health






/cloudfront-us-east-1.images.arcpublishing.com/gray/DFXC4VVIYJHHZLYDRKPUJI7CBA.jpg)